Complete Guide To Cleaning Facilities in Australia

We have spent years refining how our teams handle medical facility cleaning across Australia, and the reality is that healthcare environments sit in a category entirely separate from any other commercial space. Our crews deal with airborne pathogens, blood-borne viruses, multi-drug-resistant organisms, and vulnerable patient populations every single shift. When we first began offering medical cleaning services in Sydney, the gap between what most commercial cleaners were doing and what healthcare facilities actually needed was enormous. This guide captures everything we have learned about closing that gap and keeping clinical environments genuinely safe.
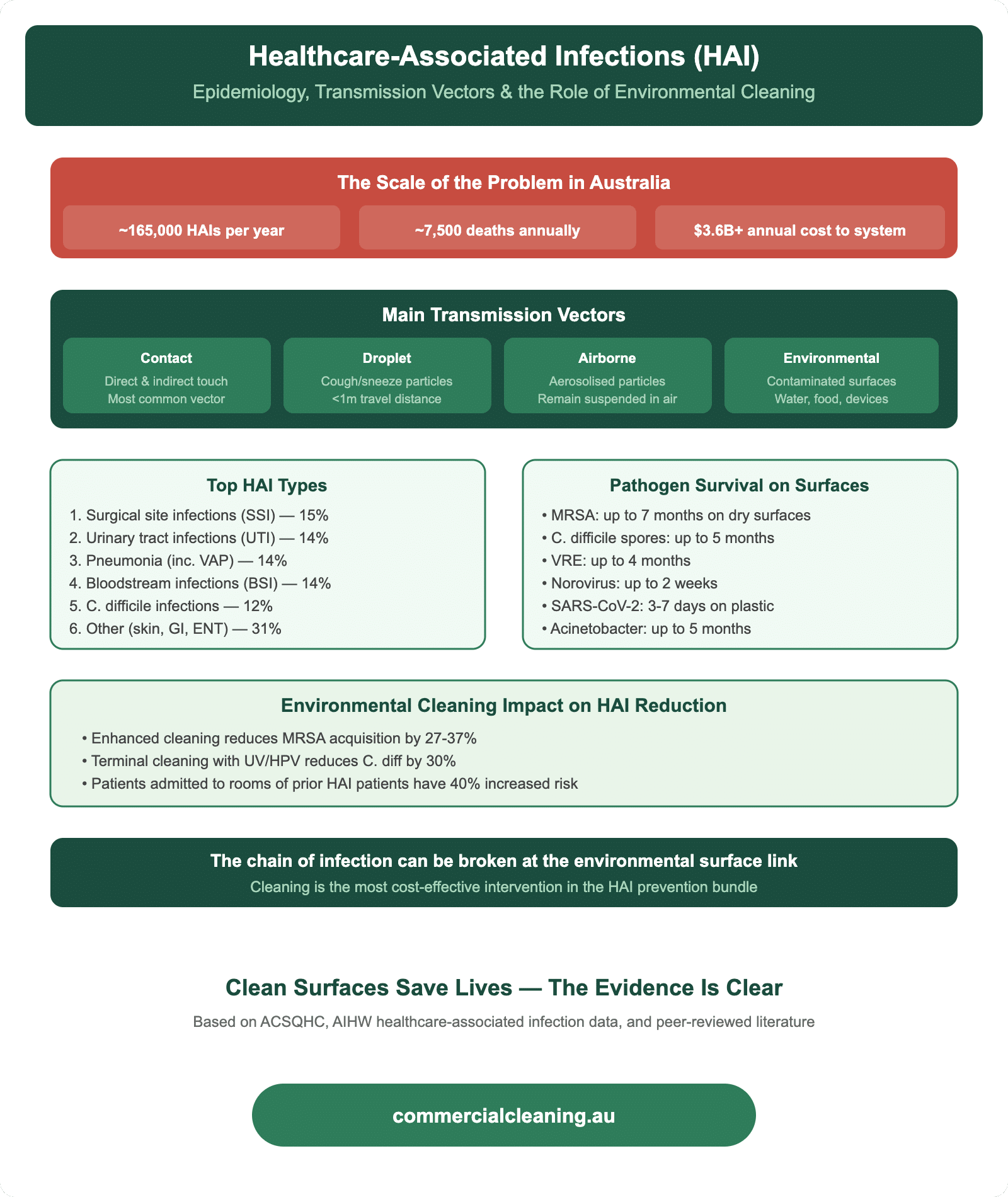
Healthcare-Associated Infection Epidemiology and Transmission Vectors
We track healthcare-associated infection data closely because it shapes every protocol we write. In our experience, environmental surfaces account for a significant share of HAI transmission, particularly with organisms like Clostridioides difficile, vancomycin-resistant Enterococcus, and methicillin-resistant Staphylococcus aureus. These pathogens survive on dry surfaces for days or even weeks, which means every surface our team misses becomes a potential transmission vector. We brief our medical cleaning crews on current HAI trends at the start of every contract because the threat landscape shifts constantly.
Our supervisors attended an infection control conference in 2023 where epidemiologists presented data showing that patients admitted to rooms previously occupied by MRSA-positive patients had a 40 percent higher risk of acquiring the same organism if terminal cleaning was inadequate. That statistic reshaped how we approach discharge cleaning. We now allocate double the time for terminal cleans compared to routine daily maintenance, and we verify every terminal clean with ATP testing before the room is released back to nursing staff.
We also pay close attention to how pathogens move through a facility via high-touch transfer points. Our mapping exercise at a medical centre in Bonnyrigg revealed that the five highest-touch surfaces were door handles, lift buttons, reception countertops, shared computer keyboards, and bathroom tap handles. We clean those surfaces a minimum of four times per day in every medical facility we service, and that frequency increases during respiratory virus season. Understanding transmission vectors at this level is what separates competent medical cleaning from guesswork.
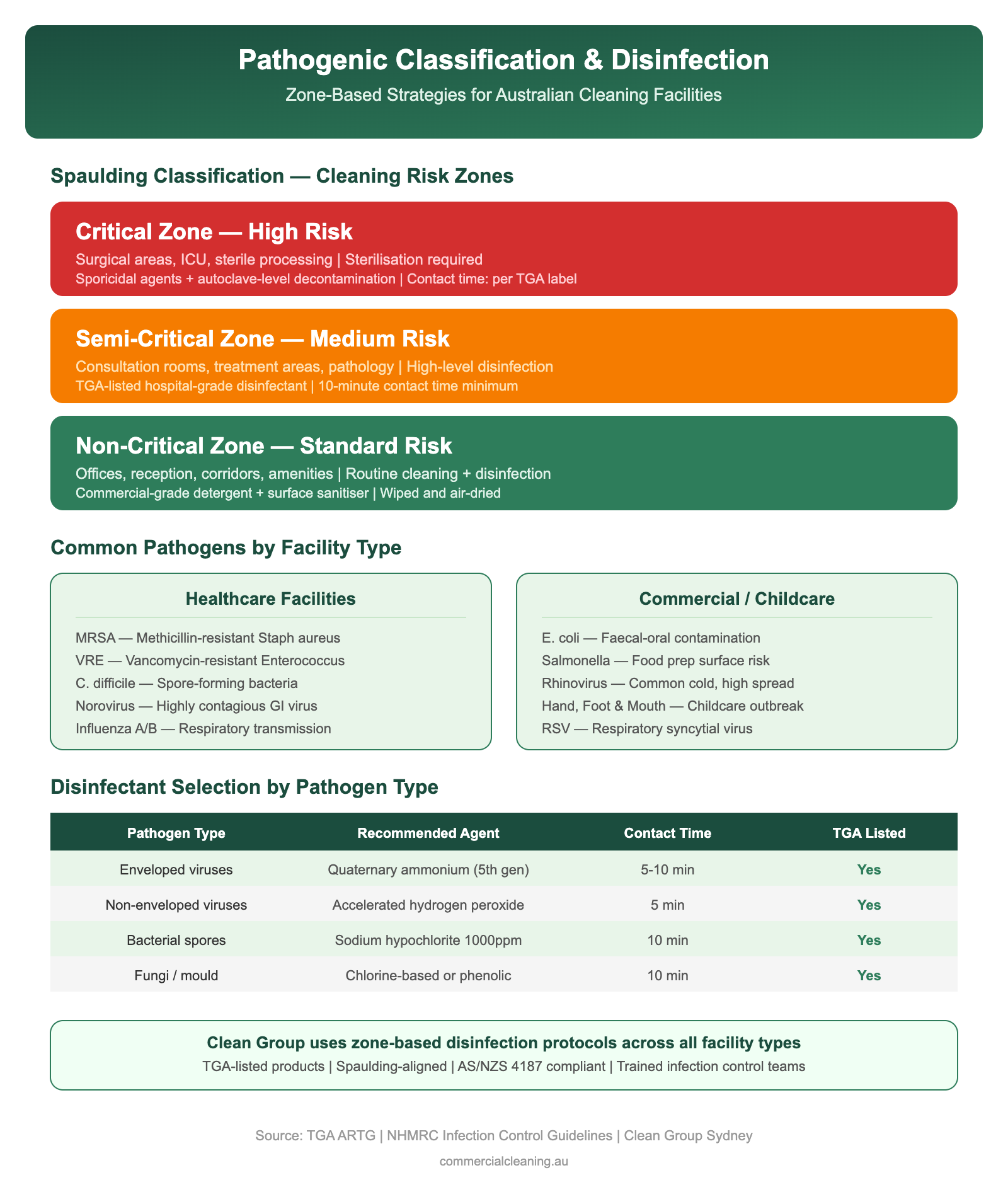
Pathogenic Classification and Zone-Based Disinfection Strategies
Pathogenic Classification and Zone-Based Disinfection Strategies involves specific protocols that we tailor to each facility based on its layout, traffic, and compliance requirements. We classify every area within a medical facility into one of four risk zones: very high risk, high risk, significant risk, and low risk. Very high risk zones include operating theatres, procedure rooms, and sterile supply departments. High risk zones cover patient treatment areas, pathology collection rooms, and wound care bays. Significant risk zones include general patient waiting areas and staff break rooms. Low risk zones are administrative offices and storage areas. Each zone gets its own disinfection protocol, chemical selection, and cleaning frequency, and our team members are trained to recognise which zone they are working in before they begin any task.
Our zone-based approach connects directly to the way we manage clinical waste streams under AS 3816, which governs the management of clinical and related wastes in healthcare settings. We segregate waste into clinical waste, cytotoxic waste, pharmaceutical waste, and general waste at the point of generation, using colour-coded bags and rigid containers that comply with the standard. Our team at a specialist clinic in Cabramatta West identified a serious waste segregation failure during their first week on site where sharps and general waste were being mixed in the same bin. We corrected the issue immediately, installed compliant containers in every room, and trained the clinic staff on proper segregation. That intervention alone eliminated a major occupational health risk for everyone in the building.
We select disinfectants based on the target organism profile for each zone. In very high risk areas, we use sporicidal agents that achieve a six-log kill against C. difficile spores. In high risk areas, we use broad-spectrum hospital-grade disinfectants effective against enveloped and non-enveloped viruses. Our chemical inventory costs roughly $1,380 per quarter across a typical mid-size medical facility, and we review product efficacy data every six months to confirm we are using the most current formulations. We never lock into a single product brand because antimicrobial resistance patterns change and our chemical choices need to keep pace.
Healthcare Cleaning Risk Zone Comparison
| Zone | Risk Level | Frequency | Disinfectant Grade | PPE Required |
|---|---|---|---|---|
| Operating Theatre | Critical | Between every case | Hospital-grade TGA | Full gown, gloves, mask |
| Patient Ward | High | 2× daily + discharge | Hospital-grade TGA | Gloves, apron |
| Waiting Room | Medium | 3× daily | Commercial-grade | Gloves |
| Admin Office | Low | Daily | General purpose | Gloves |
| Bathroom/Amenities | High | 4× daily minimum | Hospital-grade TGA | Gloves, apron, eyewear |
Terminal Cleaning and Patient Room Reprocessing Protocols
Healthcare Cleaning Risk Zone Comparison requires specific protocols that we tailor to each facility based on its layout, traffic, and compliance requirements. Terminal cleaning is where our medical cleaning expertise gets tested most rigorously. When a patient with a known or suspected multi-drug-resistant organism is discharged, our crew follows a detailed reprocessing protocol that touches every surface in the room from ceiling to floor. We start by removing all linen and bagging it separately for high-temperature laundering. We then strip disposable curtains, remove waste, and clear all personal items. Only after the room is emptied do we begin the systematic disinfection process, working from high surfaces downward and from the furthest point toward the door.
Terminal Cleaning and Patient Room Reprocessing Protocols includes specific protocols that we tailor to each facility based on its layout, traffic, and compliance requirements. We have developed a 47-point terminal cleaning checklist that our teams follow without deviation. It covers bed frames, mattress seams, overbed tables, call buttons, television remotes, window sills, light switches, power points, bathroom fixtures, soap dispensers, and every other surface a patient or visitor could have touched. Our supervisors audit completed terminal cleans using UV fluorescent marker technology, where we place invisible dots on surfaces before cleaning and then check with a blacklight afterward to confirm every marked surface was physically cleaned. Our pass rate across all medical contracts currently sits at 94 percent on first inspection, and any room below 90 percent gets re-cleaned immediately.
We introduced hydrogen peroxide vapour fogging as a supplementary step for rooms that housed patients with C. difficile or carbapenem-resistant Enterobacteriaceae. The fogging unit disperses a fine mist that reaches surfaces manual cleaning can miss, including the undersides of equipment and ventilation grilles. We invested in two portable fogging units after a particularly challenging outbreak situation at a day surgery in Canley Heights where manual cleaning alone was not achieving the microbial clearance levels the infection control team required. The fogging step added fifteen minutes to each terminal clean but brought our surface clearance rate to above 99 percent.
Flooring Systems and Equipment-Specific Disinfection
Flooring Systems and Equipment-Specific Disinfection addresses specific protocols that we tailor to each facility based on its layout, traffic, and compliance requirements. We clean a wide range of flooring types across medical facilities, and each material demands a different approach. Vinyl sheet flooring, which is the most common in clinical areas, gets damp mopped with a neutral pH detergent followed by a hospital-grade disinfectant. We never use abrasive pads on vinyl because micro-scratches harbour bacteria in their grooves and make future disinfection less effective. Polished concrete in corridors and waiting areas gets a dust mop followed by an auto-scrubber pass. Carpet tiles in administrative zones get vacuumed daily with a HEPA-filtered machine and hot water extracted quarterly.
Equipment disinfection is a separate discipline that our team takes extremely seriously. We clean examination couches, important signs monitors, infusion pump housings, and diagnostic equipment housings using manufacturer-approved wipes that are compatible with the device materials. Our technicians carry a reference folder listing approved cleaning agents for every major medical equipment brand we encounter, because using the wrong chemical on an ultrasound probe housing or a ventilator panel can degrade plastic components and void warranties. We built this reference folder over years of working with biomedical engineers across our Sydney medical contracts.
We also manage the cleaning of shared diagnostic equipment like portable X-ray machines and ECG units that travel between patient rooms. These items get wiped down between every patient contact, and we maintain a cleaning log attached to each portable device. Our team in the Bonnyrigg area pioneered this log system after a facility audit revealed that mobile equipment was being moved between isolation and non-isolation rooms without documented decontamination. The log takes thirty seconds to complete and has eliminated that compliance gap entirely.
Validation Testing and Quality Assurance
Validation Testing and Quality Assurance targets specific protocols that we tailor to each facility based on its layout, traffic, and compliance requirements. We use three complementary validation methods to confirm our medical cleaning meets the standards our clients expect. ATP bioluminescence testing gives us an immediate on-site reading of organic contamination levels. We test a minimum of ten high-touch surfaces per shift and record every result in our digital quality management system. Our threshold for clinical surfaces is 100 RLU, and we have maintained a first-pass compliance rate above 96 percent for the past eighteen months. Any reading above threshold triggers an immediate re-clean and re-test, and we investigate root cause patterns monthly to identify training gaps or process weaknesses.
Our second validation method is environmental microbiological sampling, which we conduct quarterly through an independent NATA-accredited laboratory. We collect surface swabs from predetermined locations and submit them for culture analysis targeting indicator organisms. Results come back within 72 hours, and we share them with our clients alongside trend analysis showing how microbial counts have changed over time. This data gives facility managers hard evidence for their accreditation submissions and infection control committee reports.
Our third method is observational auditing, where our quality assurance team conducts unannounced site visits and scores cleaning performance against a standardised rubric. We video-record portions of the audit with client permission and use the footage in our training programme to demonstrate both correct technique and common errors. We developed this three-layer quality assurance model after recognising that no single testing method captures the full picture. ATP readings can miss certain pathogens, culture results take days to return, and observational audits are subjective without supporting data. Together, the three methods give us and our clients genuine confidence in cleaning outcomes.
We are proud of the medical cleaning standards we maintain across every facility we service. For a closer look at the products and equipment that make these results possible, we invite you to read our next guide in our medical cleaning series.
Frequently Asked Questions
Why is medical facility cleaning different from office cleaning?
Medical facilities harbour pathogens that pose direct risks to immunocompromised patients and healthcare workers. We use hospital-grade disinfectants, follow zone-based protocols, and validate results with ATP testing and microbial cultures, none of which are standard in office cleaning. Our teams receive specialised training in infection control principles, biohazard response, and clinical waste segregation that goes well beyond what commercial cleaning requires.
What is the difference between disinfection and sterilisation?
Disinfection reduces pathogen levels on surfaces to a safe threshold but does not eliminate all microorganisms. Sterilisation destroys all forms of microbial life including bacterial spores and is reserved for critical items that enter sterile body tissue. We handle disinfection of environmental surfaces and semi-critical items, while sterilisation of surgical instruments falls within the facility’s sterile services department.
How often should medical facility rooms be cleaned?
We clean occupied clinical rooms at least once daily, with high-touch surfaces addressed twice daily in acute areas. Terminal cleaning occurs after every patient discharge. Administrative areas and low-risk zones are cleaned daily. Restrooms in clinical areas receive a minimum of three cleaning rounds per shift due to the higher pathogen load from patients with active infections.
What ATP bioluminescence result indicates adequate cleanliness?
We use a threshold of 100 relative light units for clinical surfaces. Readings below 100 RLU indicate the surface has been effectively cleaned and the organic soil that shields pathogens has been removed. Non-clinical common areas operate at a 250 RLU threshold. Any reading above these levels triggers an immediate re-clean and re-test before the area is returned to service.
How do healthcare-associated infections transmit and what is environmental cleaning’s role?
Healthcare-associated infections transmit through direct contact with contaminated surfaces, airborne droplets, and contaminated equipment. Environmental cleaning breaks the transmission chain by removing or killing pathogens on surfaces before they can transfer to patients or staff. Studies show that patients admitted to inadequately cleaned rooms face significantly higher infection risk, which is why our terminal cleaning protocols include verification testing to confirm pathogen clearance.
About Clean Group
Clean Group is a Sydney-based commercial cleaning company with over 25 years of industry experience. Founded by Suji Siv, our team of 50+ trained professionals services offices, warehouses, medical centres, schools, childcare facilities, retail stores, gyms, and strata properties across Sydney, Melbourne, and Brisbane.
We are active members of ISSA and the Building Service Contractors Association of Australia (BSCAA). Our operations align with ISO 9001 (Quality Management), ISO 14001 (Environmental Management), and ISO 45001 (Workplace Health and Safety) standards. We hold membership with the Green Building Council of Australia and use eco-friendly, TGA-registered cleaning products wherever possible.
Every Clean Group cleaner is police-checked, fully insured, and trained in safe work procedures under SafeWork NSW guidelines. We operate 7 days a week, including after-hours and weekend services, to minimise disruption to your business.


