Medical Facility Cleaning Standards in Australia
Medical cleaners sydney professionals need to understand this. We work within the Australian healthcare cleaning standards framework every day, and understanding these standards is necessary for any facility manager who wants to confirm their cleaning provider is delivering genuine compliance rather than just surface-level results. Our healthcare facility cleaning teams are trained to meet every applicable standard, from NHMRC infection control guidelines to state-based health department requirements. This guide walks through the key standards that govern how healthcare facilities should be cleaned in Australia and how we apply them in practice across the medical centres, clinics, and day hospitals we service.

Australian Standards and Regulatory Framework
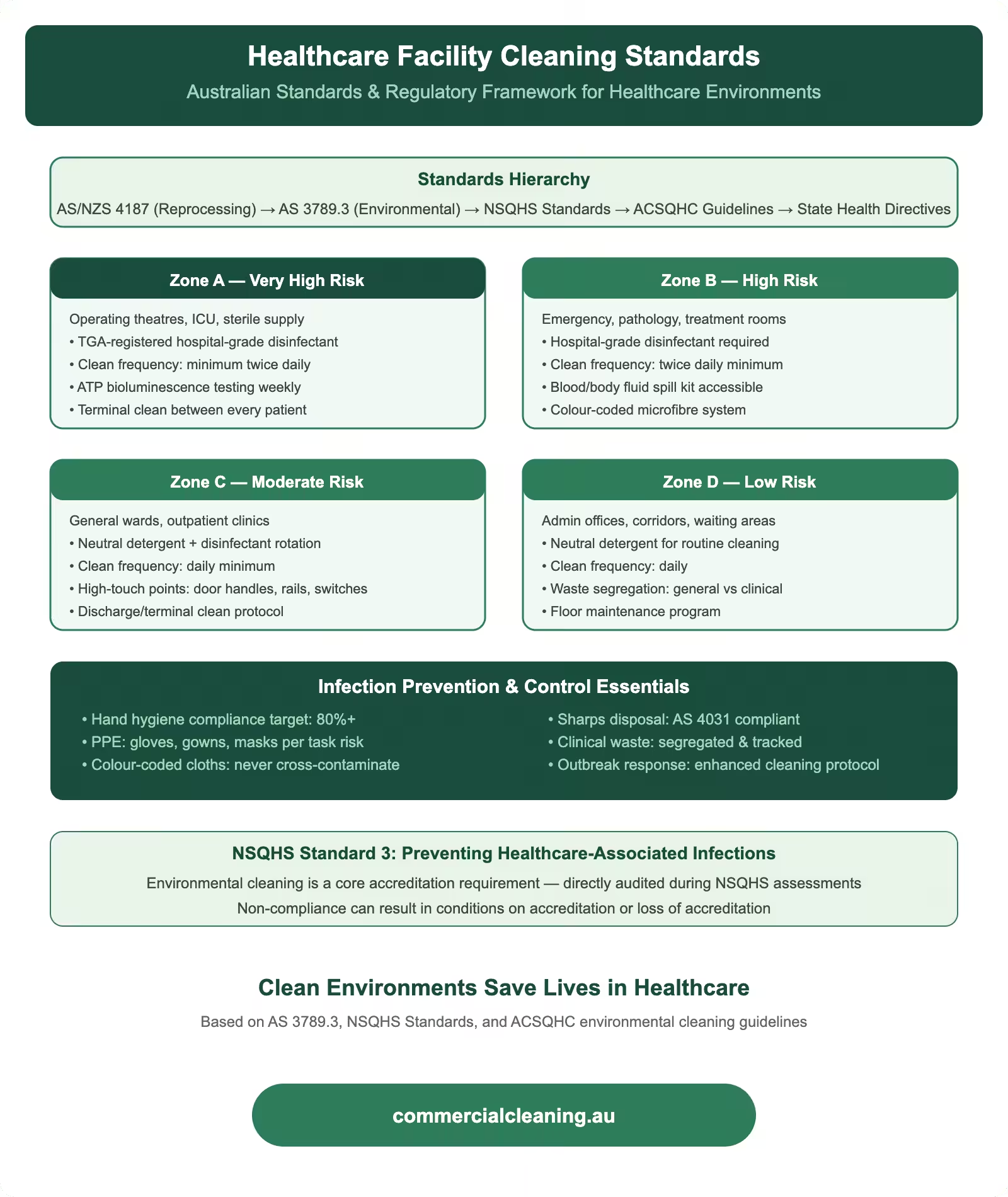
We handle a multi-layered regulatory framework that includes federal guidelines from the NHMRC, state-based health department policies, and facility-specific accreditation requirements under the National Safety and Quality Health Service standards. The NHMRC Australian Guidelines for the Prevention and Control of Infection in Healthcare provide the overarching framework that defines how cleaning fits within the broader infection prevention strategy. These guidelines establish that environmental cleaning is a critical component of standard precautions and must be performed to a level that minimises the risk of pathogen transmission from surfaces to patients, staff, and visitors.
The NSQHS Standards, administered by the Australian Commission on Safety and Quality in Health Care, include specific requirements for infection prevention and control that directly affect cleaning operations. Standard 3 covers preventing and controlling healthcare-associated infections, and it requires facilities to demonstrate that their environmental cleaning programmes are evidence-based, monitored for effectiveness, and continuously improved. We help our clients meet these requirements by providing ATP testing data, microbiological culture results, observational audit scores, and training documentation that assessors can review during accreditation visits.
We also work within the requirements of AS 4187.2, which covers the cleaning, disinfection, and sterilisation of reusable medical devices using washer-disinfectors. While the standard primarily applies to sterile processing departments, the principles of validated cleaning cycles and documented reprocessing apply to our environmental cleaning work as well. Our cleaning processes follow the same logic: pre-clean to remove organic soil, apply the correct level of disinfection for the surface risk category, validate the outcome through testing, and document every step. A medical centre we service in Toongabbie adopted this parallel approach after their infection control committee recognised that environmental cleaning should be held to the same evidence standards as instrument reprocessing.
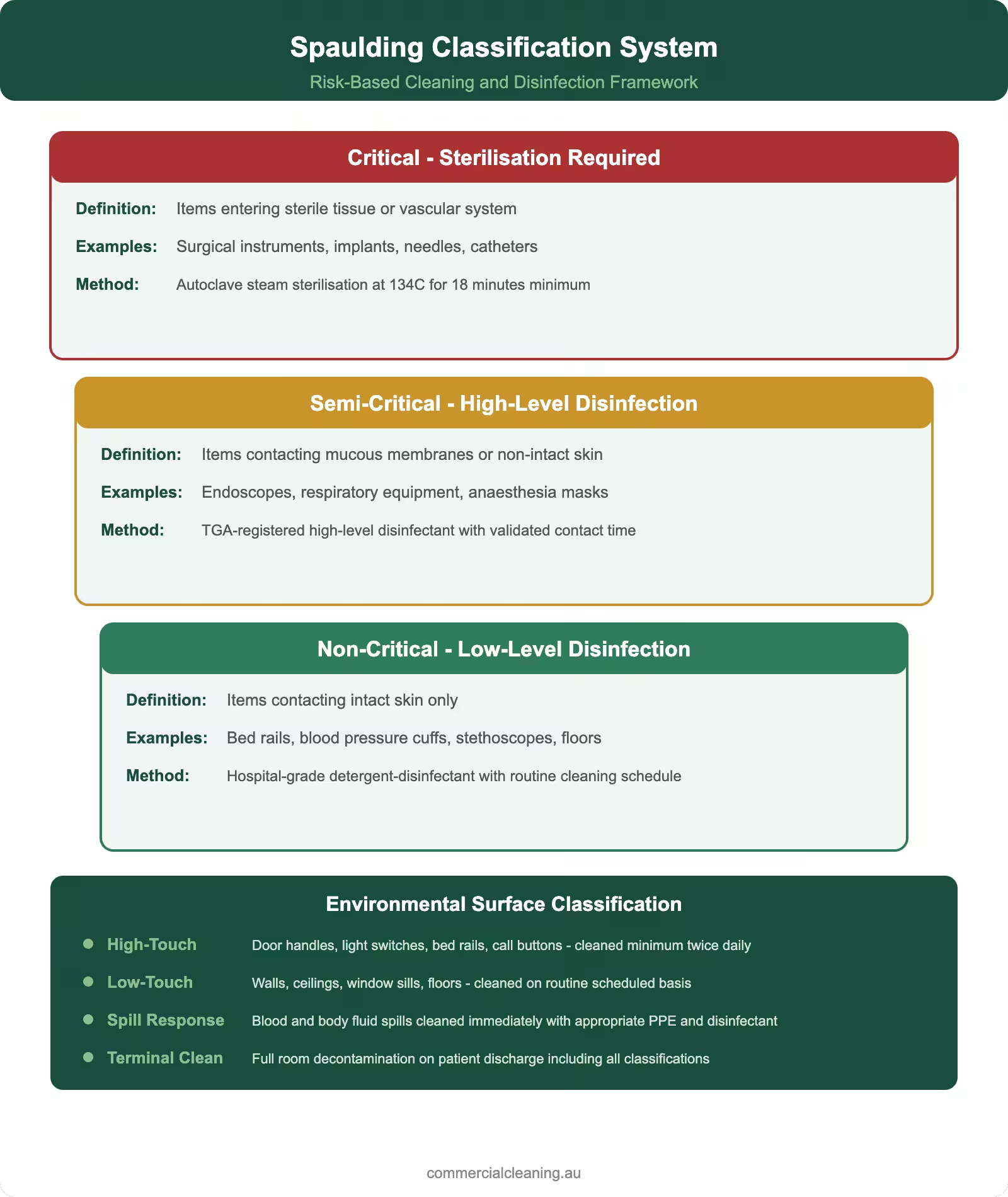
Spaulding Classification and Risk-Based Cleaning
We apply the Spaulding Classification system as the foundation of our risk-based cleaning approach in every healthcare facility. This system categorises items and surfaces into three levels: critical items that enter sterile tissue and require sterilisation, semi-critical items that contact mucous membranes or non-intact skin and require high-level disinfection, and non-critical items that contact intact skin and require low-level disinfection. While environmental cleaning primarily deals with non-critical surfaces, our teams understand the full classification system because they need to recognise when a surface has been contaminated by contact with semi-critical or critical items and escalate their disinfection response accordingly.
Our risk-based approach means we never apply a uniform cleaning protocol across an entire facility. Procedure rooms where minor surgery or invasive diagnostics occur get a higher level of disinfection than administrative offices in the same building. Patient waiting areas get more frequent touch-point cleaning than storage rooms. Isolation rooms housing patients with known multi-drug-resistant organisms get sporicidal disinfection that general patient rooms do not need. We build a customised cleaning matrix for every facility that maps the Spaulding risk level, cleaning frequency, chemical selection, and PPE requirements for every room and surface type. This matrix is reviewed annually and updated whenever the facility changes its service profile.
We have found that the most common compliance gap in healthcare cleaning is the failure to escalate disinfection levels when circumstances change. A standard examination room that normally receives low-level disinfection needs to be escalated to high-level treatment if a patient with a suspected multi-drug-resistant infection has been seen in that room. Our teams are trained to check with reception staff about patient isolation precautions at the start of every shift, and we flag any rooms that require escalated treatment on our digital task management system so nothing gets missed.
Healthcare Cleaning Risk Zone Comparison
| Zone | Risk Level | Frequency | Disinfectant Grade | PPE Required |
|---|---|---|---|---|
| Operating Theatre | Critical | Between every case | Hospital-grade TGA | Full gown, gloves, mask |
| Patient Ward | High | 2× daily + discharge | Hospital-grade TGA | Gloves, apron |
| Waiting Room | Medium | 3× daily | Commercial-grade | Gloves |
| Admin Office | Low | Daily | General purpose | Gloves |
| Bathroom/Amenities | High | 4× daily minimum | Hospital-grade TGA | Gloves, apron, eyewear |
Colour-Coded Systems and Hospital-Grade Disinfectant Standards
Colour-Coded Systems and Hospital-Grade Disinfectant Standards includes specific protocols that we tailor to each facility based on its layout, traffic, and compliance requirements. We implement a four-colour cleaning system that aligns with Australian healthcare best practice and is specifically designed to prevent cross-contamination between different risk zones within a facility. Blue cloths and mops handle general clinical areas including consultation rooms and corridors. Red handles all bathroom and toilet surfaces. Green covers kitchens, staff break rooms, and food preparation areas. Yellow is reserved exclusively for isolation rooms and areas with known or suspected infectious patients. This colour-coding extends to our mop buckets, cleaning trolley compartments, and laundry bags so every element of the cleaning supply chain maintains zone integrity from start to finish.
Our disinfectant products meet hospital-grade registration requirements under the TGA ARTG, which means they have been independently tested and validated for efficacy against specific healthcare-relevant organisms. We select products based on their activity spectrum, contact time, material compatibility, and staff safety profile. Our chemical expenditure on hospital-grade products across a typical mid-size healthcare facility runs approximately $2,080 per quarter, which reflects the higher-grade formulations and more frequent application rates compared to commercial-grade alternatives. We review our product portfolio every six months against the latest efficacy data and adjust our selections when new formulations offer improved performance or safety advantages.
We source our hospital-grade products through verified Australian distributors and maintain Safety Data Sheets for every chemical on every contract site. Our dilution control systems use calibrated dosing equipment rather than manual measurement to eliminate concentration variability. We have seen facilities where the previous cleaning provider was manually diluting concentrated disinfectant with inconsistent results, sometimes too weak to achieve the labelled kill claims and sometimes too strong, causing surface damage and unnecessary staff chemical exposure. Our standardised dilution approach eliminates both risks and ensures every application meets the product’s validated performance parameters.
ATP Testing and Environmental Monitoring Standards
We use ATP bioluminescence testing as our primary objective measure of cleaning effectiveness, aligned with the growing recognition among Australian healthcare regulators that visual inspection alone is insufficient to verify surface cleanliness. Our ATP testing protocol covers a minimum of ten high-touch surfaces per facility per shift, with results expressed in relative light units and compared against our thresholds of 100 RLU for clinical surfaces and 250 RLU for non-clinical areas. Every result is logged digitally with a timestamp, room identifier, surface type, and staff member name, creating a complete audit trail that accreditation assessors can review.
We supplement ATP testing with quarterly environmental microbiological sampling conducted through an independent NATA-accredited laboratory. Surface swabs are collected from twenty predetermined locations and submitted for culture analysis targeting indicator organisms including Staphylococcus aureus, coliforms, and Pseudomonas species. Culture results give us a more specific picture of which organisms are present on surfaces and whether our disinfection protocols are achieving adequate kill rates for the target pathogens in each facility. We share these results with our clients in a standardised report format that includes trend analysis and comparison against published benchmarks.
Our environmental monitoring programme at a specialist centre in Old Toongabbie identified a recurring Pseudomonas contamination pattern on bathroom tap handles that was not captured by ATP testing alone because the organism count was low enough to produce acceptable RLU readings but still represented a clinically significant presence. The microbiological culture results triggered an investigation that traced the source to a biofilm deposit inside the tap aerator. We reported this to the facility maintenance team who replaced the aerators, and subsequent sampling confirmed the contamination was eliminated. This example demonstrates why we use multiple monitoring methods rather than relying on any single testing approach.
Terminal Cleaning, Outbreak Response, and Documentation
We perform terminal cleaning after every patient discharge in clinical settings, following a detailed protocol that covers all surfaces from ceiling to floor. Our terminal cleaning checklist contains between 28 and 47 items depending on the room type, and our supervisors verify completion using UV fluorescent marker technology. We place invisible dots on predetermined surfaces before cleaning and check with a blacklight afterward to confirm physical contact was made with every marked point. Any missed surface triggers an immediate re-clean before the room is released for the next patient. Our terminal cleaning pass rate across all healthcare contracts sits above 94 percent on first inspection.
Outbreak response cleaning follows an escalated protocol that increases chemical strength, extends contact times, and adds supplementary disinfection steps. When a facility declares an outbreak of C. difficile, norovirus, or a multi-drug-resistant organism, we switch the affected zones to sporicidal disinfection using sodium hypochlorite at 5,000 to 10,000 parts per million depending on the organism. We increase cleaning frequency in common areas, add extra hand hygiene dispenser checks, and implement enhanced terminal cleaning that includes hydrogen peroxide vapour fogging in high-risk rooms. Our outbreak response protocol is pre-agreed with each facility during onboarding so we can activate it immediately without negotiation when a situation arises. A facility in Constitution Hill activated our outbreak protocol during a gastroenteritis cluster last winter and the environmental transmission chain was broken within 72 hours of our escalated cleaning commencing.
We are committed to helping healthcare facilities meet and exceed Australian cleaning standards. For more detailed information on medical facility standards and practices, read our next guide on essential health tips for medical facilities.
Frequently Asked Questions
What is the Spaulding classification?
The Spaulding Classification categorises medical items and surfaces into three risk levels: critical items entering sterile tissue that need sterilisation, semi-critical items contacting mucous membranes needing high-level disinfection, and non-critical items contacting intact skin needing low-level disinfection. We use this system to determine the appropriate cleaning and disinfection protocol for every surface in a healthcare facility.
What is ATP testing in healthcare cleaning?
ATP bioluminescence testing measures adenosine triphosphate on surfaces, which indicates the presence of organic matter and living cells. We express results in relative light units and use thresholds of 100 RLU for clinical surfaces and 250 RLU for non-clinical areas. Results are logged digitally for audit trails and trend analysis that support accreditation reviews.
What is hospital-grade disinfectant?
Hospital-grade disinfectants are registered on the TGA Australian Register of Therapeutic Goods and have been independently tested for efficacy against specific healthcare pathogens including Pseudomonas aeruginosa and Staphylococcus aureus. We only use ARTG-registered products in healthcare facilities because they provide validated kill claims that commercial-grade alternatives cannot guarantee.
What is a terminal clean in healthcare?
A terminal clean is a detailed cleaning and disinfection process performed after a patient is discharged, covering all surfaces from ceiling to floor. We use a detailed checklist of up to 47 items and verify completion with UV fluorescent marker technology. Terminal cleans are more thorough than routine daily cleaning and are required for breaking infection transmission chains between patients.
How do Australian healthcare cleaning standards support accreditation?
The NSQHS Standards require healthcare facilities to demonstrate evidence-based environmental cleaning programmes. We support accreditation by providing ATP testing data, microbiological culture results, observational audit scores, documented cleaning logs, and training competency records. This documentation package demonstrates systematic, monitored, and continuously improved cleaning practices that assessors specifically evaluate during accreditation visits.
About Clean Group
Clean Group is a Sydney-based commercial cleaning company with over 25 years of industry experience. Founded by Suji Siv, our team of 50+ trained professionals services offices, warehouses, medical centres, schools, childcare facilities, retail stores, gyms, and strata properties across Sydney, Melbourne, and Brisbane.
We are active members of ISSA and the Building Service Contractors Association of Australia (BSCAA). Our operations align with ISO 9001 (Quality Management), ISO 14001 (Environmental Management), and ISO 45001 (Workplace Health and Safety) standards. We hold membership with the Green Building Council of Australia and use eco-friendly, TGA-registered cleaning products wherever possible.
Every Clean Group cleaner is police-checked, fully insured, and trained in safe work procedures under SafeWork NSW guidelines. We operate 7 days a week, including after-hours and weekend services, to minimise disruption to your business.




