Infection Control Cleaning Standards for Medical Practices

Every medical practice that prioritises strong infection control cleaning standards depends on disciplined sydney medical centre cleaning routines, and we have seen firsthand how those routines protect patients.
We have spent countless hours studying how surfaces behave in medical environments, and the insights we have gathered fundamentally changed the way our healthcare cleaning teams approach every shift. Spotless surfaces in a medical facility are not just about appearance — they are a direct line of defence against healthcare-associated infections that can put vulnerable patients at serious risk. Our approach to medical surface cleaning combines scientific understanding of biofilm behaviour, objective measurement tools, and practical cleaning techniques refined through years of hands-on work in clinics, day surgeries, and specialist centres across Sydney.
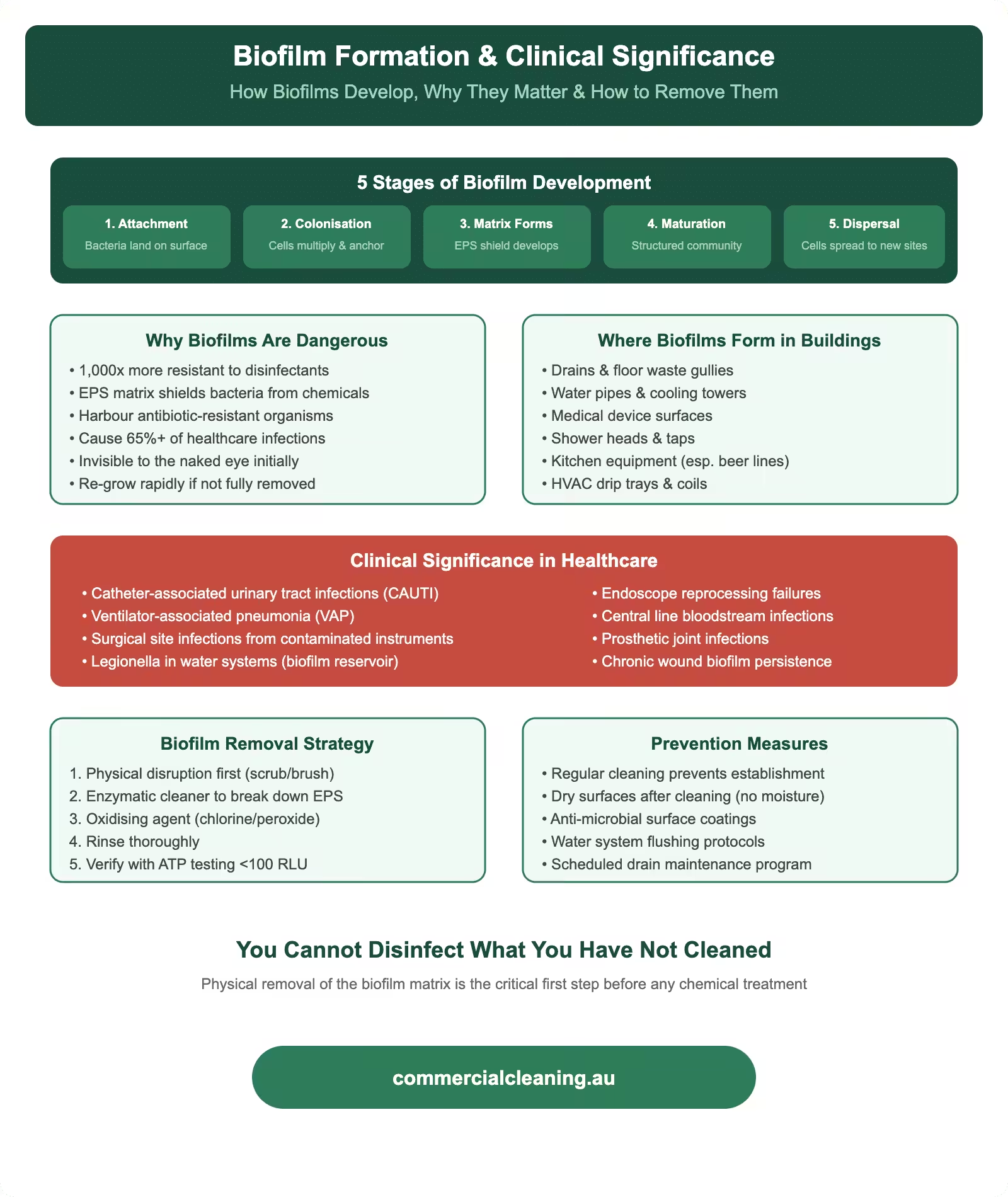
Biofilm Formation and Its Clinical Significance
Biofilm Formation and Its Clinical Significance covers specific protocols that we tailor to each facility based on its layout, traffic, and compliance requirements. We pay close attention to biofilm because it represents one of the most persistent challenges in medical surface cleaning. Biofilm forms when microorganisms attach to a surface and produce an extracellular matrix that shields them from both the immune system and disinfectant chemicals. In our experience, biofilm begins forming within hours on surfaces that are not properly cleaned, and once it matures, it becomes extraordinarily difficult to remove with standard wiping alone. We have tested surfaces in medical centres that appeared visibly clean but harboured mature biofilm colonies detectable only through ATP testing or microbiological sampling.
Our approach to biofilm prevention centres on two principles: mechanical removal through proper wiping technique and chemical disruption using enzymatic or oxidative cleaners that break down the biofilm matrix. We train our crews to use firm, overlapping strokes rather than light passes when wiping surfaces, because the physical friction is what dislodges the biofilm layer. We then follow with a hospital-grade disinfectant that can penetrate any residual matrix. This two-step approach consistently produces ATP readings below 100 RLU on surfaces that previously tested above 500 RLU under the old contractor’s single-step wipe method.
We encountered a particularly stubborn biofilm problem at a specialist consultation suite in Strathfield South where the stainless steel bench surfaces in the procedure room were returning elevated microbial counts despite daily disinfection. Our investigation revealed that the surfaces had micro-scratches from years of abrasive cleaning that created perfect harbourage sites for biofilm. We recommended the facility resurface those benches and switched their cleaning protocol to a non-abrasive enzymatic pre-cleaner followed by accelerated hydrogen peroxide. The microbial counts dropped to undetectable levels within two weeks and have stayed there since.

ATP Bioluminescence Testing for Objective Surface Measurement
We adopted ATP bioluminescence testing as our primary surface cleanliness verification tool because it gives us an objective number within seconds rather than relying on visual inspection alone. A surface can look perfectly clean to the naked eye while carrying enough organic residue to sustain pathogenic growth. Our handheld ATP meters detect adenosine triphosphate, which is present in all living cells and organic matter, and express the result in relative light units. We set our clinical surface threshold at 100 RLU and our non-clinical threshold at 250 RLU, and we test a minimum of ten surfaces per facility per shift.
We log every ATP result into our digital quality management platform where it is timestamped, geotagged to the specific room, and linked to the team member who performed the clean. This data allows us to identify patterns over time — whether a particular surface type consistently produces higher readings, whether certain shifts have lower compliance rates, or whether a specific cleaning product is underperforming. We review aggregated ATP data monthly with our quality assurance team and share quarterly trend reports with each medical facility we service. Our clients use these reports as evidence in their NSQHS accreditation reviews and RACGP infection control audits.
We calibrate our ATP meters monthly at our depot and replace the units annually to confirm accuracy. We also run positive and negative control tests at the start of each batch of swabs to verify the reagent is performing correctly. This level of rigour in our testing methodology gives us and our clients confidence that every ATP result is meaningful and actionable. We have seen other providers wave an ATP meter around as a marketing gimmick without the calibration and control testing to back up their numbers, and that approach undermines the value of the technology entirely.
Healthcare Cleaning Risk Zone Comparison
| Zone | Risk Level | Frequency | Disinfectant Grade | PPE Required |
|---|---|---|---|---|
| Operating Theatre | Critical | Between every case | Hospital-grade TGA | Full gown, gloves, mask |
| Patient Ward | High | 2× daily + discharge | Hospital-grade TGA | Gloves, apron |
| Waiting Room | Medium | 3× daily | Commercial-grade | Gloves |
| Admin Office | Low | Daily | General purpose | Gloves |
| Bathroom/Amenities | High | 4× daily minimum | Hospital-grade TGA | Gloves, apron, eyewear |
Microfibre Technology and Colour-Coded Cleaning Systems
Healthcare Cleaning Risk Zone Comparison requires specific protocols that we tailor to each facility based on its layout, traffic, and compliance requirements. We transitioned our entire medical cleaning division to microfibre cloths and flat mops over five years ago, and the improvement in cleaning outcomes was immediate and measurable. Independent studies show that microfibre removes up to 99 percent of microorganisms from surfaces with water alone, compared to around 30 percent removal with traditional cotton cloths. We still apply disinfectant for the chemical kill step, but the mechanical removal capability of microfibre means the disinfectant has far less organic matter to penetrate, which dramatically improves its effectiveness. Our teams use pre-dampened microfibre cloths from sealed bags to verify consistent moisture levels across every wipe.
Microfibre Technology and Colour-Coded Cleaning Systems includes specific protocols that we tailor to each facility based on its layout, traffic, and compliance requirements. Our colour-coded cloth system prevents cross-contamination between zones. We use blue cloths for general clinical surfaces, red for bathroom and toilet areas, green for kitchen and staff break rooms, and yellow for isolation and high-risk clinical zones. Each cloth is single-use within a zone and gets bagged separately for laundering at the end of each clean. We enforce this system without exception because cross-contamination from using a bathroom cloth on a treatment room surface defeats the entire purpose of disinfection. Our laundry process thermally disinfects all microfibre at temperatures above 70 degrees Celsius to eliminate any residual pathogens before the cloths are reissued.
We also maintain strict rules about cloth folding technique. Our teams fold each microfibre cloth into quarters, which gives them eight usable cleaning faces per cloth. They work through each face systematically rather than scrunching the cloth and wiping randomly. This technique maximises the cleaning surface area, distributes disinfectant evenly, and prevents the recontamination that occurs when a soiled section of cloth contacts a clean surface. We assess cloth folding technique during our monthly competency observations, and it is one of the skills that distinguishes a properly trained medical cleaning technician from a general commercial cleaner.
Surface-Specific Disinfection Protocols
We tailor our disinfection approach to each surface material because using the wrong product or technique on the wrong surface creates problems ranging from reduced disinfection efficacy to permanent material damage. Stainless steel surfaces in procedure rooms get wiped with a neutral pH quaternary ammonium product that does not leave streaks or corrode the finish. Laminate countertops in consultation rooms get a similar treatment but we avoid products containing bleach because it degrades the laminate seal over time. Vinyl flooring in clinical areas gets damp mopped with a hospital-grade floor disinfectant applied at the label dilution rate, and we never use wax-based polishes because they create a film that harbours bacteria in its surface imperfections.
Electronic equipment requires particular care. We clean computer screens, keyboards, tablets, and touchscreens using alcohol-based wipes that are compatible with electronic components and approved by the equipment manufacturer. Our product reference guide lists the recommended cleaning agent for every major medical equipment brand we encounter. We developed this guide after an incident at a day procedure centre in Enfield where a cleaning technician used a bleach-based wipe on an infusion pump touchscreen that permanently damaged the display coating. The reference guide eliminated that category of error from our operations entirely.
Our handling of single-use medical items aligns with the principles outlined in AS 4179, which covers single-use infusion equipment standards. While the standard itself deals with manufacturing specifications, the underlying principle that single-use items must not be reprocessed applies directly to our work. We never attempt to clean or reuse items marked as single-use, including disposable curtain tracks, single-patient blood pressure cuff covers, and disposable speculum handles. Our teams are trained to identify the single-use symbol and dispose of these items correctly. The cost of replacing single-use items, approximately $2,150 per quarter across a typical medical facility, is far less than the infection control risk of attempting to reprocess them.
Environmental Monitoring and Continuous Improvement
We run a three-layer environmental monitoring programme across all our medical facilities. The first layer is ATP bioluminescence testing that gives us immediate feedback on surface cleanliness. The second layer is quarterly environmental microbiological sampling through an independent NATA-accredited laboratory, where we collect surface swabs from twenty predetermined high-touch points and submit them for culture analysis. The third layer is air quality sampling in procedure rooms and sterile supply areas, where we measure particulate counts and viable airborne organisms to make sure ventilation systems and our cleaning practices are maintaining acceptable air quality standards.
We use the data from all three monitoring layers to drive continuous improvement in our cleaning protocols. When ATP trend data shows a gradual increase in readings across a facility, we investigate whether the cause is a product issue, a technique issue, or a staffing issue. When microbiological culture results identify an unusual organism on a surface, we cross-reference with the facility’s clinical records to determine whether it represents a community pathogen brought in by patients or a potential environmental persistence issue. Our Croydon Park team identified an emerging VRE presence on bathroom surfaces at a medical centre through routine sampling and helped the facility implement enhanced disinfection protocols before any patient transmission occurred.
We present our monitoring data to clients in a standardised dashboard format that tracks key metrics over time. The dashboard shows ATP compliance rates, microbial culture results, audit scores, and incident reports alongside industry benchmarks. Several of our medical centre clients have told us this dashboard was instrumental in their successful NSQHS accreditation assessments because it demonstrated a systematic, evidence-based approach to environmental cleaning that the assessors specifically praised. We invest considerable effort in making our data accessible and actionable because we believe transparency is the foundation of a strong cleaning partnership.
We are passionate about raising the bar for medical surface cleaning across every facility we service. For necessary tips on cleaning frequency schedules in medical centres, we recommend exploring our next guide in our medical cleaning series on medical centre cleaning frequency.
Frequently Asked Questions on Medical Surface Cleaning
What does an ATP reading below 250 RLU indicate for medical surface cleanliness?
An ATP reading below 250 RLU on a non-clinical surface indicates that organic contamination has been effectively removed. For clinical surfaces in treatment and procedure rooms, we use a stricter threshold of 100 RLU. Readings below these thresholds confirm that the surface has been cleaned thoroughly enough for the subsequent disinfection step to work effectively against pathogens.
How does biofilm formation reduce antimicrobial disinfectant efficacy?
Biofilm produces an extracellular matrix that acts as a physical and chemical barrier between the disinfectant and the microorganisms embedded within it. This matrix can reduce disinfectant penetration by up to 1,000-fold compared to free-floating organisms. We address this by using mechanical wiping to physically disrupt the biofilm layer before applying disinfectant, which allows the chemical to reach and kill the organisms underneath.
Why is organic matter removal critical before chemical disinfection?
Organic matter including blood, body fluids, skin cells, and dust creates a physical barrier that prevents disinfectant from contacting the surface and the pathogens on it. Many disinfectants are also chemically neutralised by organic material, which reduces their effective concentration below the kill threshold. We always perform a detergent pre-clean to remove organic soil before applying disinfectant because skipping this step can reduce disinfection effectiveness by 60 percent or more.
How should colour-coded microfibre cloths be used to prevent cross-contamination?
We assign each cloth colour to a specific zone: blue for general clinical areas, red for bathrooms, green for kitchens and break rooms, and yellow for isolation and high-risk zones. Each cloth is used only within its designated zone and disposed of after a single use. Cloths are folded into quarters to provide eight clean faces, and our teams work through each face systematically to maximise cleaning efficiency and prevent recontamination.
What environmental monitoring validates medical facility cleaning effectiveness?
We use three complementary methods: ATP bioluminescence testing for immediate surface cleanliness feedback, quarterly microbiological culture sampling through an independent laboratory for pathogen identification, and air quality monitoring in procedure rooms for particulate and viable organism counts. Together these three layers provide detailed evidence that our cleaning protocols are delivering the infection control outcomes medical facilities require.
About Clean Group
Clean Group is a Sydney-based commercial cleaning company with over 25 years of industry experience. Founded by Suji Siv, our team of 50+ trained professionals services offices, warehouses, medical centres, schools, childcare facilities, retail stores, gyms, and strata properties across Sydney, Melbourne, and Brisbane.
We are active members of ISSA and the Building Service Contractors Association of Australia (BSCAA). Our operations align with ISO 9001 (Quality Management), ISO 14001 (Environmental Management), and ISO 45001 (Workplace Health and Safety) standards. We hold membership with the Green Building Council of Australia and use eco-friendly, TGA-registered cleaning products wherever possible.
Every Clean Group cleaner is police-checked, fully insured, and trained in safe work procedures under SafeWork NSW guidelines. We operate 7 days a week, including after-hours and weekend services, to minimise disruption to your business.


